Bellwork
1. Convert 350 0C to K
2. Describe the particle attraction and kinetic energy in solids, liquids, and gases.
3. If a gas has a decreased pressure by 1/2, how will the volume be changed?
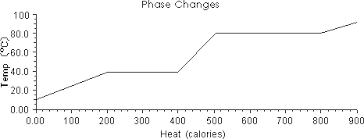
4. In the graph below determine the melting point, the boiling point, and how much energy is required to boil the substance.
1. Convert 350 0C to K
2. Describe the particle attraction and kinetic energy in solids, liquids, and gases.
3. If a gas has a decreased pressure by 1/2, how will the volume be changed?
4. In the graph below determine the melting point, the boiling point, and how much energy is required to boil the substance.
Classwork
TEST Ch. 10/11 Kinetic Energy and Gas Laws
Begin Ch. 13 Properties of Water (handout)
TEST Ch. 10/11 Kinetic Energy and Gas Laws
Begin Ch. 13 Properties of Water (handout)
 RSS Feed
RSS Feed
